|
Therefore, the whole procedure of DNA extraction and PCR is prone to reagent contamination. Conventional (not real time) PCR and subsequent electrophoresis are time-consuming as well, particularly since two rounds of long-lasting nested PCR are performed. Manual DNA extraction is time-consuming and a potential source of contamination of processed clinical specimens. Although real time PCR was developed many years ago, the application of 16S rDNA (including melting curve analysis for the differentiation of specific and nonspecific PCR products) came much later, probably because of the restriction of the maximal size of amplified fragments in real time PCR and because replacement of existing methods usually takes time. For a long time, manual DNA extraction and non-real time amplification with subsequent electrophoretic separation of PCR products remained the state of the art. The first reports in diagnostic microbiology starting at the end of the nineteen eighties describe the use of highly hazardous chemicals for the extraction of nucleic acids and detection of PCR products. For this analysis in particular, the available methods have changed during the past 30 years. Very few results at the detection limit still have to be critically examined.įor the identification of bacterial species and/or genera, 16S rDNA PCR is a standard method in most clinical microbiology laboratories. Clinical specimens with truly PCR-positive but culture-negative results represent an additional benefit for patients. 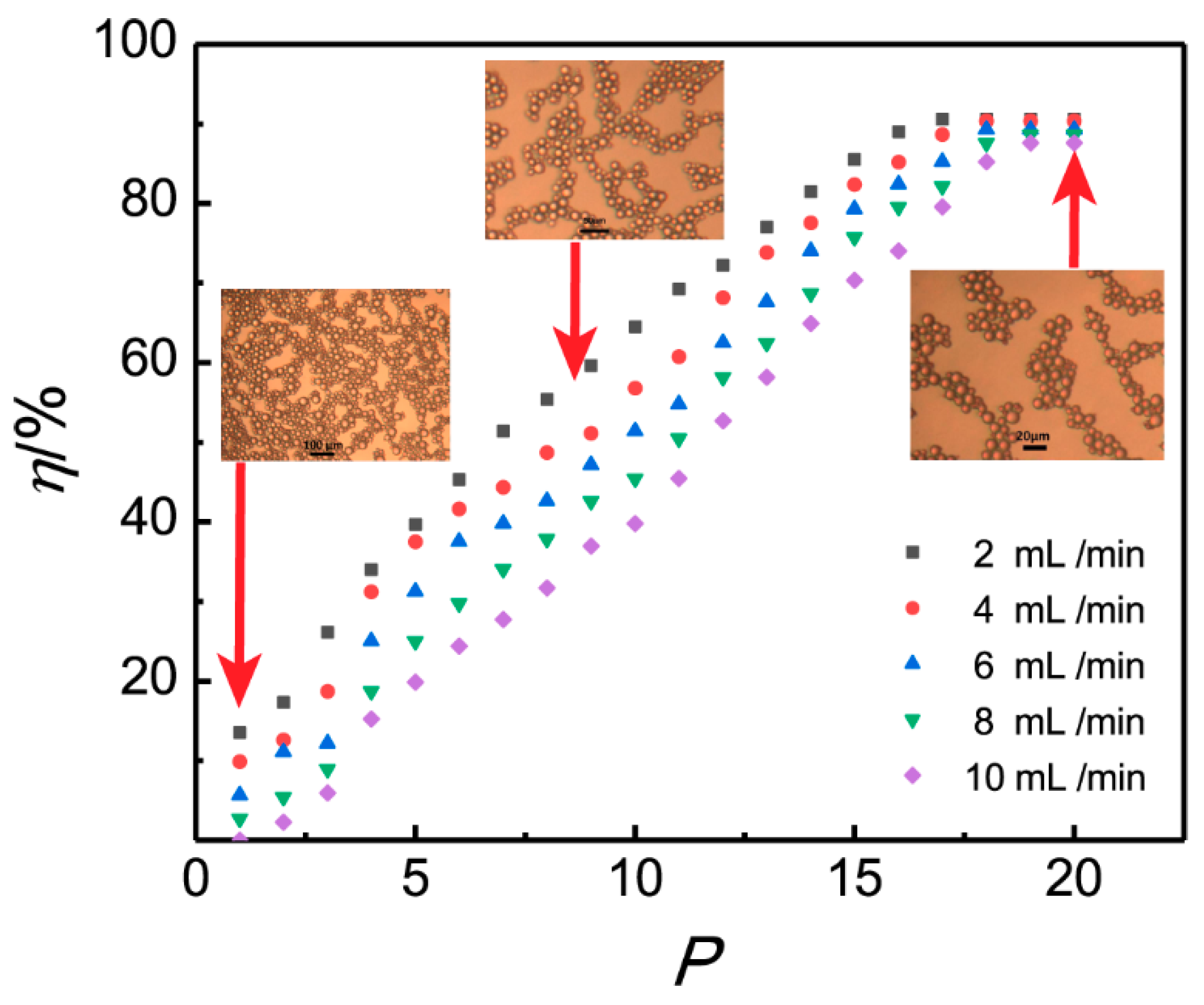
The crucial innovations of the technology enable timely reporting of explicit reliable results for adequate treatment of patients. Conclusionsįor endocarditis cases, the added benefit of PCR is obvious. Using Ripseq, even mixed infections of 2 bacterial species could be resolved. Melting-curve analysis of SYBR Green-PCR enables the differentiation of specific and unspecific PCR products.  
The turnaround times for negative specimens were 4 hours (automated DNA extraction and real time PCR) and 1 working day for positive specimens (including Sanger sequencing). Thirty-eight (4.2%) clinical specimens were culture-positive (25 of them were only enrichment culture positive) but PCR-negative, mainly for S. Many of these 62 positive specimens were strongly positive based on crossingpoint values (54% < Cp 30), and these 62 positive clinical specimens were diagnosed as medically relevant as well. A total of 170 (21.2%) clinical specimens were PCR-positive, of which 62 (36.5% from PCR positive, 7.7% in total) gave an additional benefit to the patient since only the PCR result was positive. In 595 (74.1%) samples, the results were concordant negative, and in 102 (12.7%) the results were concordant positive. The clinical specimens were mainly of orthopedic origin received at our diagnostic laboratory. In total, we compared 803 clinical specimens using real time PCR and culturing. Here, we describe the results obtained with a new technology that uses DNA-free reagents for automated DNA extraction and subsequent real time PCR using sterile clinical specimens. Additionally, previously used methods are frequently time-consuming. However, the DNA extraction reagents as well as the PCR reagents may contain residual bacterial DNA, which consequently generates false-positive PCR results. 
16S rDNA-PCR for the identification of a bacterial species is an established method.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
